How does the cord blood process and store once collected?
Absolutely Safe and Painless
for Both Mother and Infant
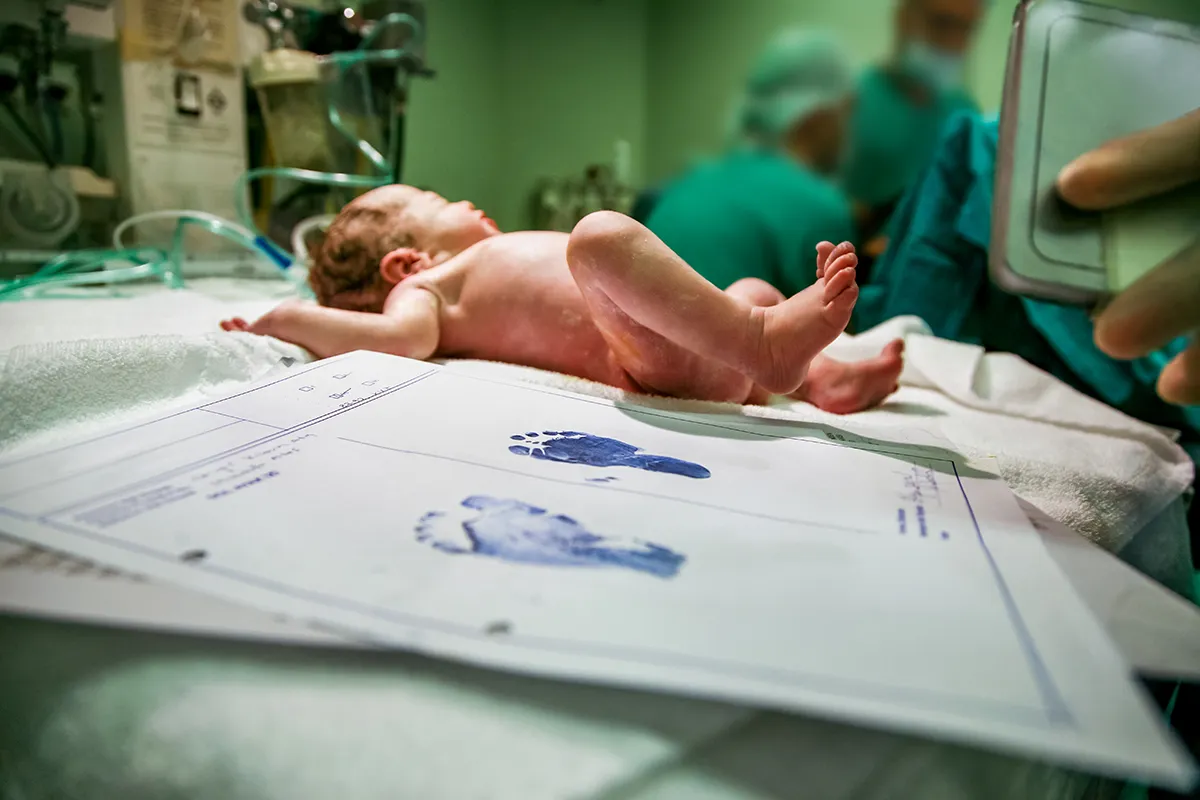
Cord Blood is collected after the infant leaves the mother’s body and the umbilical cord is cut
Collection Procedure of Cord Blood
Cord Blood is collected after the infant leaves the mother’s body and the umbilical cord is cut, so the process of cord blood collection is absolutely safe and painless for both mother and infant. The quantity of cord blood depends on the size of the placenta and the thickness of the umbilical cord, it about 60ml on average for Asian. Every single drop of cord blood is rich in stem cells, so doctors will endeavor to collect as much cord blood as possible.
1
Cut & Sterilize
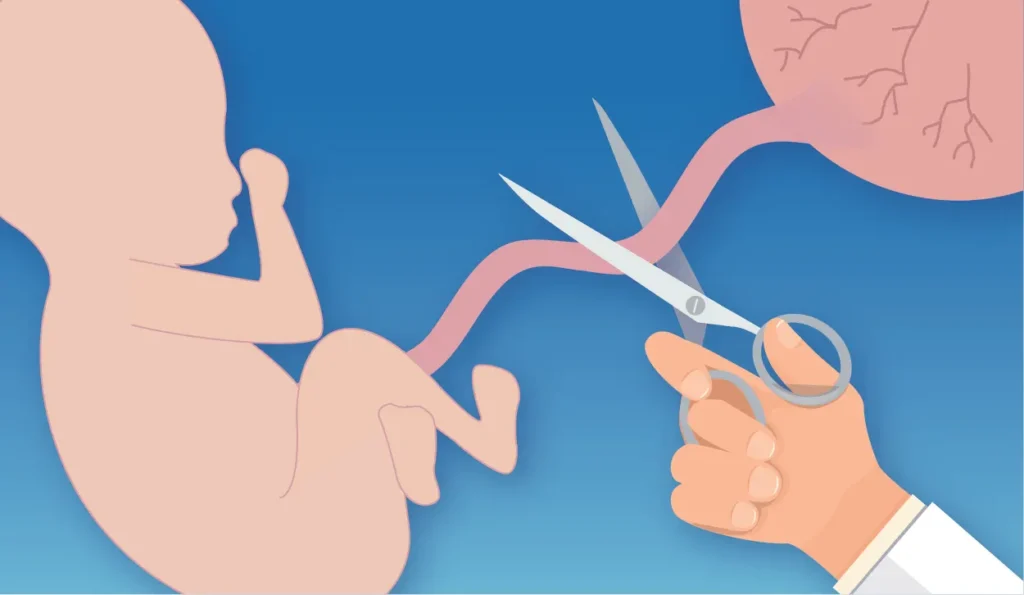
Cut and sterilize the needle insertion point of umbilical cord 3 times after baby is born.
2
Collecting Cord Blood
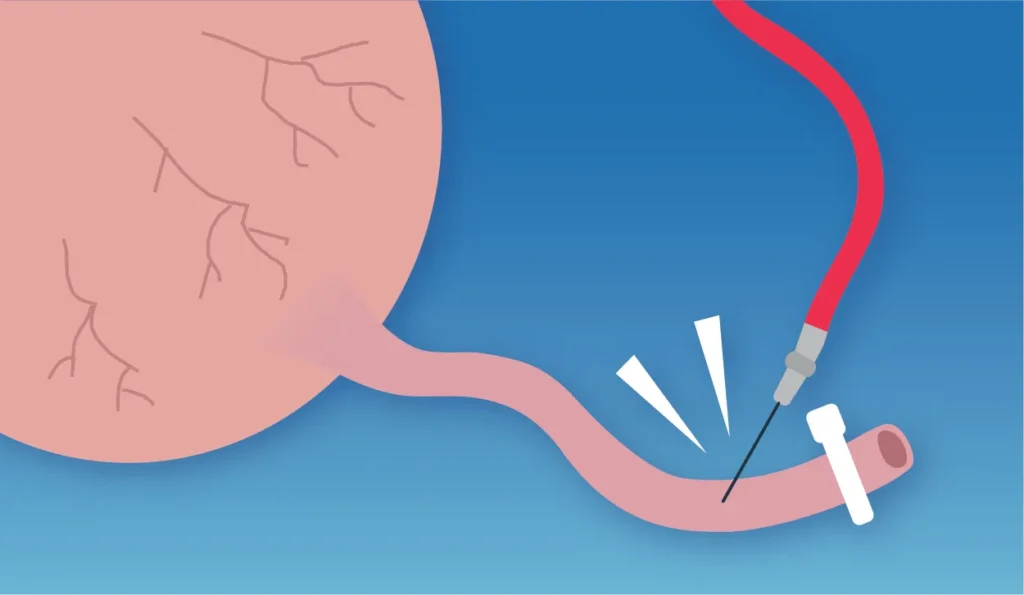
Insert the needle at the vein near the end of the umbilical cord, collect blood quickly (to be completed in 5-10 minutes) to avoid coagulation. Squeeze out all the blood in the placenta and umbilical cord until the umbilical cord looks pale.
3
Collection Bag Sealing
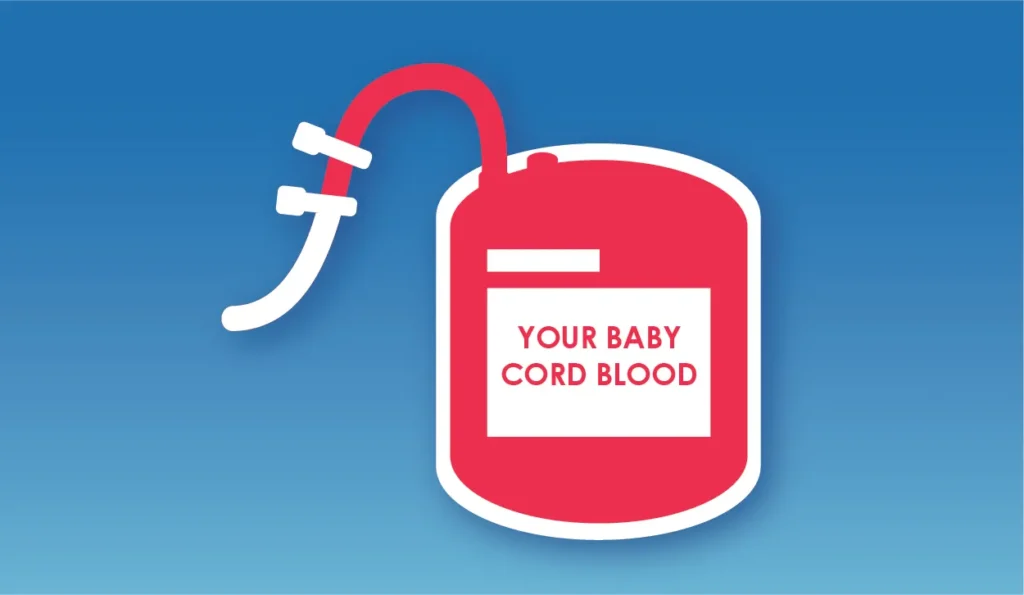
After collection, use two “cord clips” to seal the site 5-7cm away from the opening of the blood bag to prevent any leakage or pollution of the cord blood.
Cord Blood Processing Procedure 1
We will assign dedicated staff to collect the cord blood after delivery
Baby’s cord blood will be placed inside the freezing box (15 -25℃) and our dedicated staff will deliver the cord blood from hospital to laboratory for processing. Please call our hotline after delivery.
Cord Blood Processing Procedure 2
Identification & sample testing
Identity information will be verified and an unique ISBT barcode will be assigned for detailed record and international tracking. Next, Laboratory techinician will test the cord blood according to international accreditation requirement to ensure the cord blood is free from ontamination and stem cells viability is suitable for storage.
Cord Blood Processing Procedure 3
Stem Cells Isolation using AXP® cord blood processing system
Once confirmed the cord blood is suitable for storage, cord blood would be loaded into AXP® System to segregate white blood cells and red blood cells, normally will take around 45mins for this procedure.
Cord Blood Processing Procedure 4
Temperature control and storage
Insert the canister into controlled rate freezer. Once the designated temperature is reached, the canister will be put into BioArchive® System for cryogenic storage.
Cord Blood Processing Procedure 5
Lab report & all time monitoring
Comprehensive laboratory report will be sent by post after baby is born (8 weeks). Should you have any enquiries, please feel free to call us at our hotline.
HealthBaby will regularly release Quality Check Report to ensure clients’ stem cells reaching the transplant standard. (Laboratory results are tested from laboratory samples)
- HealthBaby routinely conducts annual quality assurance test according to AABB and CAP guidelines to closely monitor the quality of stem cells stored. Cord blood reference samples preserved in BioArchive® System with different storage duration have been thawed to evaluate the viability of stem cells.
Most Asked Questions by Parents Related to Cord Blood Banking
Until today, up to 113 diseases had been treated with cord blood transplants5. They could be divided into 7 categories, namely: Leukemia, Lymphomas, other Disorders of Blood Cell Proliferation, Inherited Disorders of Immune System, Inherited Metabolic Disorders, other Cancers and others.
Cord blood and umbilical blood contain two different stem cells which are widely adopted in different medical aspects. Cord blood is a rich source of HSCs which can create the blood and immune cells. Cancers, blood-related, immunity and metabolic diseases can be treated by cord blood. Umbilical cord is a rich source of MSCs which can create structural and connective tissues. Apart from minimizing GvHD in directed and unrelated allogeneic cord blood transplants, MSCs can potentially apply to chronic illnesses treatment, regenerative medicines as well as organ and tissue engineering5,6. Since cord blood and umbilical cord are rich sources of different types of stem cells, they may help to repair the body in different ways.
Entrusted by most parents in Hong Kong1, HealthBaby has a very comprehensive service coverage. Apart from the traditional liquid nitrogen storage plan, HealthBaby also pioneered to provide the advanced BioArchive® System. All storage plan of HealthBaby adopts the expensive liquid nitrogen as storage medium to provide the most stable tempertature for stem cell viability, providing the most quality umbilical cord and cord blood storage service.
To cater for different needs of clients, HealthBaby's cord blood and umbilical cord storage plan have very flexible and comprehensive service plan. Regarding cord blood plan, clients could select the storage plan base on the storage and processing system needs.
The umbilical cord and cord blood service fee already includes registration fee, processing fee and 18 years storage fee, no any additional charge during the contracted year. Extra discounted would be provided for registering both umbilical cord and cord blood storage plan. HealthBaby existing customers and other cord blood bank customers could also enjoy special offers. Please contact us for deatils and any enquiries.
For more details of cord blood service plan please refer to Service Plan.
More Questions?
Contact us through any method below:
Hotline: (+852)3188-8899 / (+853)2878-6717
WhatsApp: (+852)9660-8271 or Click Here
WeChat Official Account: HealthBaby 生寶臍帶血庫
Online: Click here to submit the enquiry online
Email: enquiry@healthbaby.hk